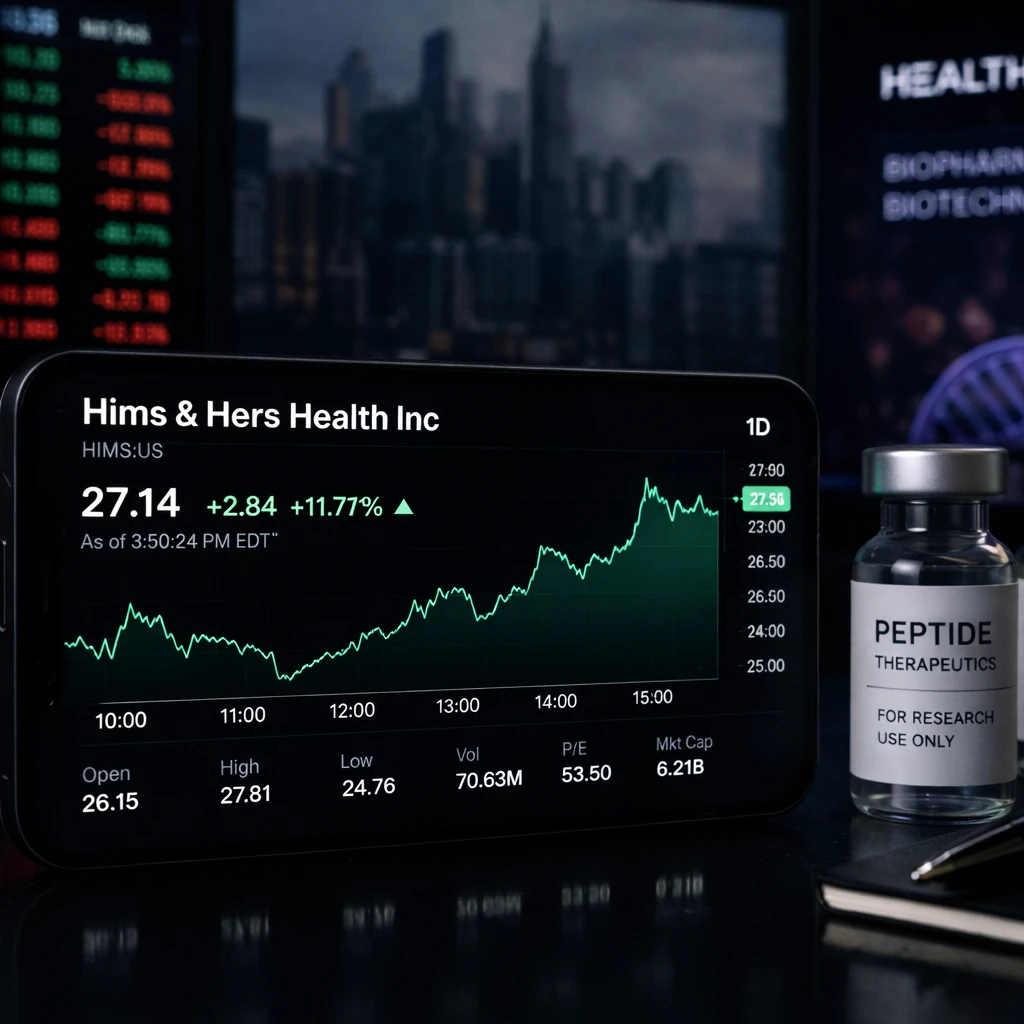
Hims & Hers Health (NYSE: HIMS) surged sharply in Thursday trading, climbing to $27.14, up +11.71% intraday, as investors reacted to a fresh regulatory development that could reshape the future of peptide-based wellness treatments.
Trading activity surged alongside the price move, with volume reaching over 70 million shares, nearly double its average daily volume of around 35 million. The stock jumped from a previous close of $24.29 and briefly touched an intraday high near $27.80, reflecting strong investor interest following the FDA update.
The rally follows news that the U.S. Food and Drug Administration has begun a formal review process for several restricted peptide compounds, a move that could open new long-term opportunities for companies operating in the compounding and telehealth space.
FDA review sparks new growth narrative
The FDA confirmed it will hold advisory committee meetings on July 23–24 to evaluate whether certain peptide substances should be included on the 503A bulk drug list. Compounds under review include BPC-157, TB-500, MOTs-C, semax, and epitalon, all of which are currently restricted in various forms.
This development does not immediately change regulatory rules, but it introduces a potential pathway for future approval or expanded compounding use. The agency has also opened a public comment window ahead of the review process, signaling early-stage regulatory consideration rather than a finalized decision.
Wall Street reacts with cautious optimism
Following the announcement, Bank of America raised its price target on Hims & Hers to $25 from $21, while maintaining a neutral stance. The revision reflects rising sector valuation multiples and growing interest in peptide-related therapies.
Analysts emphasized that this is not a near-term earnings catalyst but rather a long-term optionality driver. The outcome of the FDA’s review remains uncertain, especially regarding whether these compounds will be approved for compounding or broader commercial use.
According to FDA regulatory updates, such advisory committee reviews typically represent early procedural steps before any formal regulatory changes are implemented.
Business impact tied to existing infrastructure
For Hims & Hers, the potential upside lies in its existing compounding capabilities. If regulatory pathways open up, the company could leverage its current infrastructure—originally scaled for GLP-1 and weight-loss treatments—to expand into new peptide-based categories.
This could help improve operational efficiency by spreading fixed costs across a broader product portfolio, strengthening margins over time if demand materializes.
However, uncertainty remains a key factor. Analysts note that the regulatory path is still evolving, and any commercial opportunity will depend on future FDA decisions following the July review meetings.
Stock momentum meets valuation reality
Despite the strong rally, HIMS currently trades with a PE ratio of 53.5x and a market cap of approximately $6.2 billion, suggesting that expectations for future growth are already priced into the stock.
The company’s high beta of 2.32 also indicates elevated volatility, meaning sharp price swings—both upward and downward—are likely to continue as new regulatory updates emerge.
Investors are increasingly viewing Hims & Hers as a platform positioned at the intersection of telehealth, wellness, and emerging pharmaceutical trends, but the timeline for monetizing peptide-related opportunities remains uncertain.
For now, the FDA review has injected fresh momentum into the stock, with markets reacting to the possibility of a much larger addressable market in the years ahead.