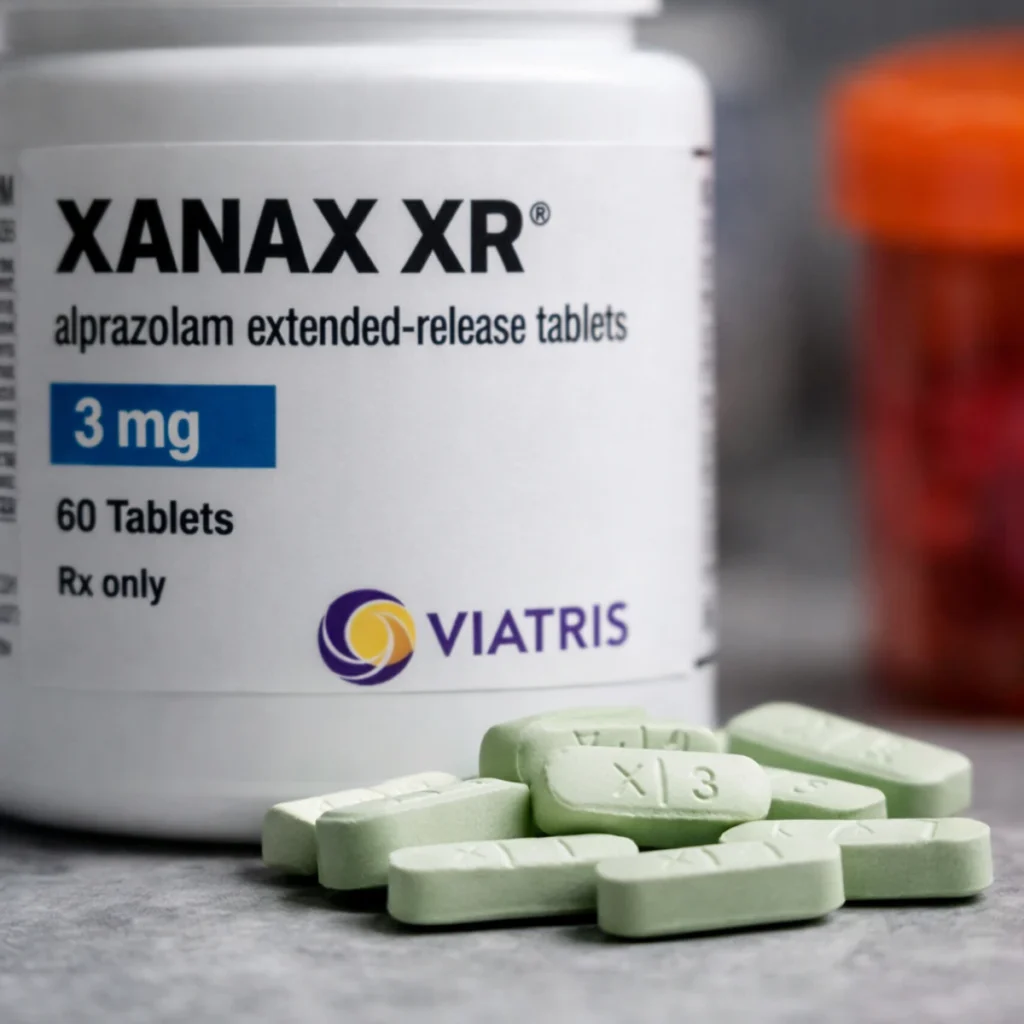
A specific batch of the widely prescribed anxiety drug Xanax XR has been recalled nationwide in the United States after failing a key quality control test, according to federal health authorities. The recall, flagged by the Food and Drug Administration (FDA), is drawing attention due to the drug’s widespread use in treating anxiety and panic disorders.
The recall affects Xanax XR (alprazolam) 3mg extended-release tablets, manufactured by Viatris Inc., with lot number 8177156. The medication was distributed across the country between August 27, 2024, and May 29, 2025, and comes in bottles of 60 tablets with an expiration date of February 28, 2027.
Regulators said the batch failed to meet “dissolution specifications,” a standard that determines how a drug releases its active ingredient in the body over time. For extended-release medications like Xanax XR, this is critical — the drug is designed to deliver a controlled dose gradually rather than all at once.
The FDA has classified the recall as a Class II event, meaning use of the affected product may lead to temporary or medically reversible adverse health effects. While not considered life-threatening, the issue raises concerns for patients relying on consistent symptom control.
Why the failure matters for patients
Failure to meet dissolution standards can result in tablets that release too little or too much of the active ingredient at the wrong time. In practical terms, patients may experience reduced effectiveness, inconsistent relief from anxiety symptoms, or unexpected fluctuations in how the medication works.
Health experts note that alprazolam is one of the most commonly prescribed benzodiazepines in the United States, making any quality-related recall significant. Because the drug is often used to manage ongoing anxiety or panic conditions, even minor inconsistencies in dosage delivery can disrupt treatment.
Unlike contamination-related recalls, this issue does not involve foreign substances or immediate toxicity. Instead, it highlights a manufacturing inconsistency that could affect how the drug is absorbed into the bloodstream, potentially altering its therapeutic effect.
What patients should check now
Patients who have been prescribed Xanax XR are being advised to check their medication packaging for the affected lot number 8177156. The product label may also include the National Drug Code (NDC) 58151-506-91 and indicate it was manufactured in Ireland and distributed by Viatris Specialty LLC.
Medical professionals caution against abruptly stopping alprazolam without guidance, as sudden discontinuation can lead to withdrawal symptoms. Patients who believe they may have the affected batch should consult their pharmacist or healthcare provider to discuss alternatives or replacements.
The recall reflects ongoing monitoring of pharmaceutical products even after they reach the market. According to the FDA’s official recalls database, such actions are taken when products may pose a risk due to defects or performance issues.
While the recall is limited to a single batch, the case underscores the importance of manufacturing consistency for medications that depend on precise dosing over time — particularly those used to manage mental health conditions where stability is critical for patients.
You may like: Tamia South Africa Tour 2026: Dates, Tickets and Venues Announced