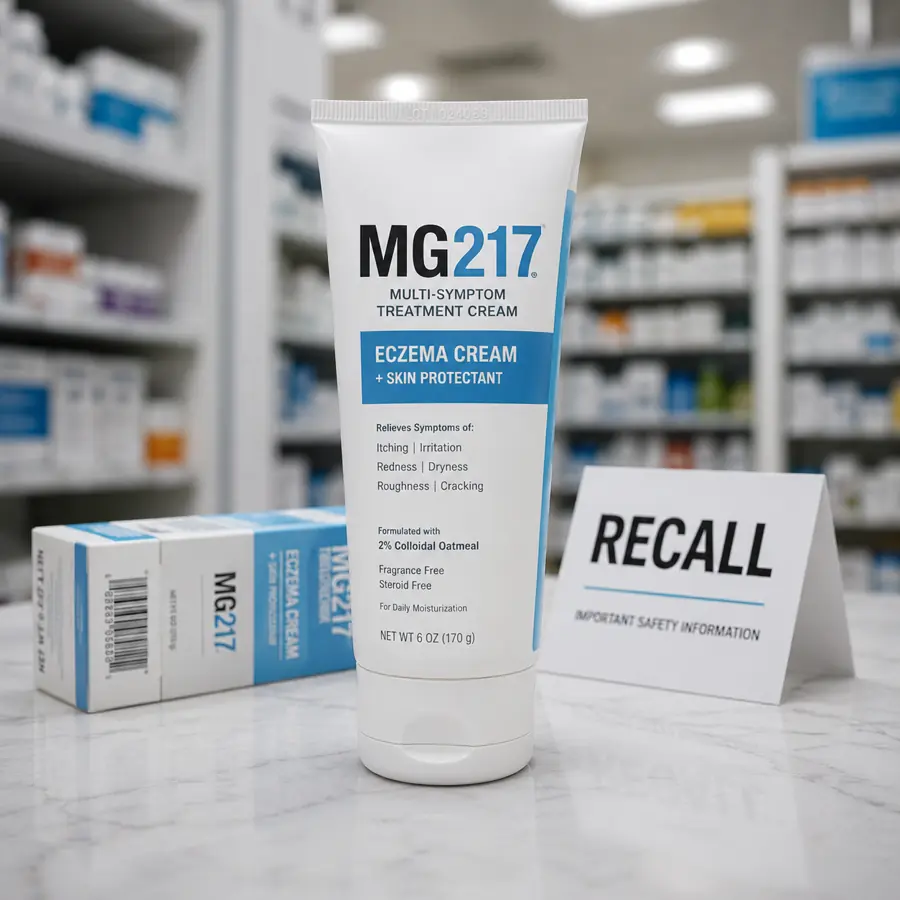
The FDA has posted a nationwide recall for MG217 Multi-Symptom Treatment Cream & Skin Protectant Eczema Cream after one lot of the over-the-counter skin product tested positive for Staphylococcus aureus, a bacteria better known for causing staph infections.
The recall was initiated by Wisconsin-based Pharmacal and affects a specific 6-ounce tube of MG217 eczema cream sold under the MG217 brand. The product was distributed across the United States through wholesale and retail channels, as well as online marketplaces including Amazon. H-E-B Grocery was also listed among retail outlets where the product was sold.
This is not a broad recall of every MG217 product. It applies to one identified lot of the eczema cream. Consumers should check the label carefully before using any tube they have at home.
Which MG217 eczema cream is recalled?
The recalled product is MG217 Multi-Symptom Treatment Cream & Skin Protectant Eczema Cream. It was sold in a 6-ounce tube and can be identified by the following details:
- Brand: MG217
- Company: Pharmacal
- Product: Multi-Symptom Treatment Cream & Skin Protectant Eczema Cream
- Product code: 5106
- UPC: 012277051067
- Lot number: 1024088
- Expiration date: November 2026
- Package size: 6 oz tube
The lot number and expiration date are printed on the crimped end of the tube. If the numbers match, consumers should stop using the cream immediately and throw it away.
The affected cream is advertised as a full-body skin protectant moisturizer for eczema symptoms. Pharmacal’s product information describes it as a treatment for itching, irritation, dryness, cracking, redness and roughness. The company has also marketed the product as hypoallergenic and safe for children and babies, which makes label checks especially important for families who keep eczema products for regular use.
The FDA said use of the contaminated cream could lead to infections ranging from localized skin problems to severe or life-threatening adverse events. That risk matters because eczema creams are often applied to inflamed, cracked or irritated skin, where the body’s natural protective barrier may already be weakened.
Why staph contamination is a serious concern
Staphylococcus aureus can live on the skin or in the nose without always causing illness, but it can become dangerous when it enters broken skin or spreads deeper into the body. In a contaminated topical cream, the concern is direct contact with already sensitive or damaged skin.
Possible infections can include skin infections, bloodstream infections, bone infections, joint infections and infections affecting the heart valves. In severe cases, staph infections can contribute to sepsis or shock, both of which require urgent medical care.
People with weakened immune systems face higher risk. So do consumers with wounds, burns, skin disorders or active eczema flare-ups. Babies, older adults and people using immune-suppressing medication should be especially careful because infections may progress faster or become harder to manage.
So far, Pharmacal has not reported any confirmed adverse events linked to the recalled lot. Still, the recall notice advises consumers not to continue using the product. A cream can look, smell and feel normal even when microbial contamination is present.
Anyone who used the recalled tube should watch for symptoms such as increased redness, swelling, warmth, pain, pus, fever, unusual drainage or skin irritation that becomes worse instead of improving. People who notice these symptoms should contact a doctor or healthcare provider, particularly if the cream was used on broken skin or on a child.
Consumers and healthcare professionals can report side effects through the FDA’s MedWatch Safety Information and Adverse Event Reporting Program. Reports help federal regulators track whether recalled products have caused illness after reaching consumers.
Read More
Pharmacal said it is notifying distributors by email and arranging the return of recalled products. Retailers may also remove affected tubes from shelves or issue notices to customers, but consumers should not wait for a store alert. The safest step is to check the product code, UPC, lot number and expiration date directly on the tube.
For households that buy skin-care items online, it is worth checking bathroom cabinets, medicine bins, diaper bags and travel kits. Eczema creams are often kept for months and may not be used every day, which means the recalled lot could remain in homes even after stores begin pulling it from sale.
This recall also shows why product codes and lot numbers matter during FDA safety alerts. A brand name alone is not always enough to identify whether a product is affected. In this case, the recall is tied to MG217 eczema cream with lot number 1024088 and an expiration date of November 2026, not necessarily every item sold under the MG217 name.
Consumers looking for another eczema product should speak with a pharmacist or healthcare provider before switching, especially when treating babies, children, severe eczema or open skin. Using a contaminated or unsuitable product on irritated skin can make symptoms worse rather than provide relief.
The FDA has posted several consumer safety alerts in recent weeks involving contamination or labeling problems across different product categories. Swikblog recently covered another FDA-linked recall involving Horizon Organic chocolate milk boxes recalled over a packaging defect, as well as a separate Spring & Mulberry chocolate recall tied to salmonella concerns.
For MG217 users, the action is straightforward: find the tube, check the lot details and stop using it if it matches the recall. If symptoms appear after use, seek medical advice and report the issue through FDA MedWatch.